Gases are found practically everywhere on Earth: in the atmosphere, dissolved in the water of the oceans, and even buried in porous solid materials. Gases play an important role in a wide range of phenomena. Indeed, most living organisms are influenced by the presence of a mixture of gases commonly known as air. Air pressure affects the activities of living organisms through variations in weather conditions.
The Earth's atmosphere is made up of a mixture of different gases. These are mainly nitrogen (78%) and oxygen (21%).
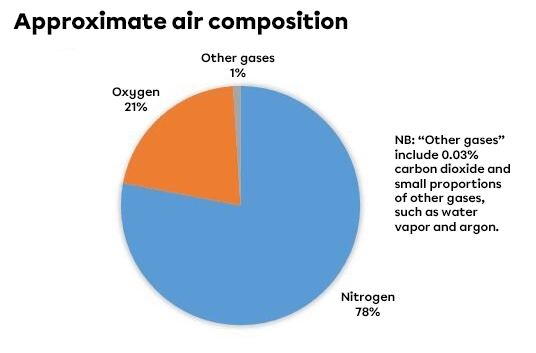
Several natural phenomena of a physical nature are generated by the various gases in the atmosphere:
- Winds correspond to the movement of air particles from an area of high pressure to an area of low pressure. It is therefore the movement of air masses that regulates the climate. Hurricanes and tornadoes are extreme manifestations of winds.
- Cloud formation is caused by the evaporation of water, and the condensation of water vapour is responsible for precipitation. Geysers project large quantities of water vapour into the atmosphere.
- Volcanoes release large quantities of gases into the atmosphere, such as water vapour (|H_{2}O|), carbon dioxide (|CO_{2}|), sulfur dioxide (|SO_{2}|) and carbon monoxide (|CO|).
- Buoyancy in fish is provided by their swim bladder, an organ that fills with oxygen (|O_{2}|) and nitrogen (|N_{2}|) extracted from the blood, enabling them to adapt to variations in water pressure.
Some examples of natural physical phenomena involving gases: volcanic eruptions, geysers, cloud formation and tornadoes.
Other phenomena involving gases are chemical in nature:
- Respiration involves the transformation of oxygen (|O_{2}|) into carbon dioxide (|CO_{2}|).
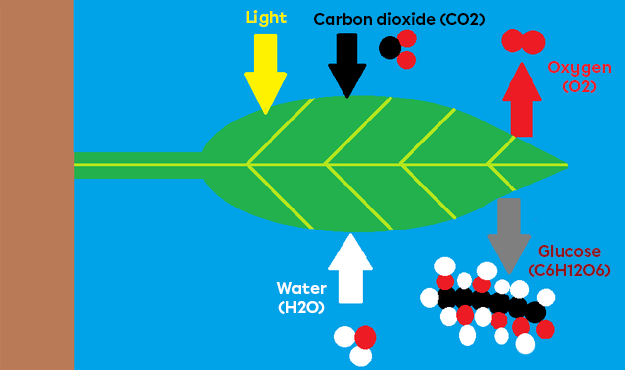
- Photosynthesis, the opposite reaction to respiration, transforms carbon dioxide (|CO_{2}|) into oxygen (|O_{2}|).
- The formation of smog, a fog made up of suspended atmospheric pollutants, involves tropospheric ozone (|O_{3}|).
- The ozone layer in the stratosphere is made up of a gas capable of absorbing ultraviolet rays from the Sun.
- Forest fires and other types of combustion release many gaseous substances into the atmosphere, mainly carbon dioxide (|CO_{2}|).
- Large quantities of methane (|CH_{4}|) are produced when permafrost heats up in the polar regions, or in low-oxygen wetlands such as marshes.
Some examples of natural chemical phenomena involving gases: photosynthesis, smog formation, forest fires and melting permafrost.


The balance of certain natural phenomena can be disturbed by an additional input of gases into the atmosphere as a result of human activity.