The cell membrane which surrounds the cell plays a very important role in cellular exchanges. With its numerous pores and membrane transporters, it is responsible for the entry and exit of all kinds of molecules, regardless of their size and nature. In general, the internal medium of a cell has a different composition from that of the external medium. Diffusion and osmosis are two of the passive mechanisms (requiring no energy) by which the cell can interact with its environment.
Pour valider ta compréhension à propos des échanges cellulaires de façon interactive, consulte la MiniRécup suivante :

Diffusion of matter is a process by which particles in a solute tend to move from an area of high concentration to an area of low concentration.
This movement takes place in order for the concentrations in both areas to be at equilibrium, in other words, for both areas to have the same concentration.
To fully understand the phenomenon, just pour ink (solute) into a glass of water (solvent). First, the solvent molecules disperse in a disorderly fashion and then collide with each other. Diffusion continues until the solute concentration is the same throughout the container. In the case of the cell, diffusion takes place across the cell membrane. For example, oxygen that enters the body through the lungs travels from the blood to the cells by means of diffusion.

The term diffusion can also refer to the scattering of waves, such as the scattering of light. This phenomenon is in no way related to cellular exchanges. In the case of light, then we would say that diffusion is the random and multi-directional deflection of light rays when they strike a solid, liquid, or gaseous body.
The phenomenon of osmosis is a process by which particles of water (or other solvent) tend to move from an area with a lower solute concentration to an area with a higher solute concentration.
This movement takes place in order for the concentrations in both areas to be at equilibrium, in other words, for both areas to have the same concentration.
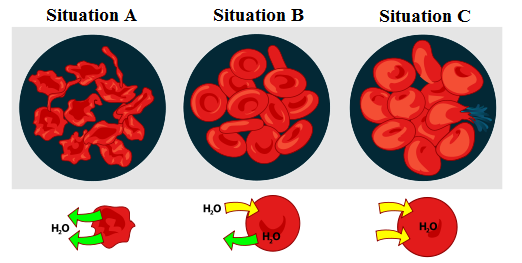
Three situations involving osmosis are possible, and they are represented in the following image. In situation A, the solute concentration is greater in the external medium than in the red blood cells; this medium can be qualified as hypertonic. To bring the situation back to equilibrium, the water inside the red blood cells will flow out of them. This is called the phenomenon of plasmolysis.
Situation B is the optimal situation, where the solute concentration is identical in the external medium and in the red blood cells. This medium can be described as isotonic.
In situation C, the solute concentration is greater inside the red blood cells than in the external medium. This medium is called hypotonic. To bring the situation back to equilibrium, water from the external medium will enter the red blood cells. This is called the phenomenon of turgescence.