The chemical reactivity of a gas is its tendency to undergo a chemical transformation under the effect of various factors such as heat, light, or contact with another substance.
The physical properties of gases can be applied to almost all gaseous substances. Therefore, they do not make it possible to identify gases since they are not characteristic. On the contrary, the chemical properties are characteristic of a particular gas or group of gases. Thus, in order to distinguish one gas from another, the chemical reactivity of gases can be used, namely how they react chemically under certain conditions.
Knowing the chemical reactivity of gases leads to several applications. Among other things, this makes it possible to identify certain gases. For example, if a gas clouds limewater, it is carbon dioxide. Also, it is possible to determine the use of gases and what technological applications can use them. For example, natural gas is used in some furnaces because it is an excellent fuel. The chemical reactivity of the gas will also determine which safety rules should be observed when handling a gas. Finally, all gases can have an asphyxiant or suffocating effect when replacing oxygen. Detectors can, therefore, be used to signal a gas concentration that is too close to the toxicity threshold.
The chemical reactivity of a gas depends mainly on the electronic configuration of atoms. More specifically, the interaction between nuclei and valence electrons is responsible for the reactivity of a gas. This electronic configuration determines whether an atom tends to gain or lose electrons.
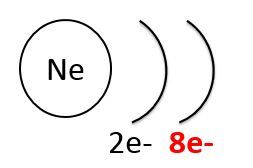
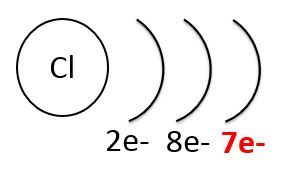
Noble gases, like neon (below to the left), have low chemical reactivity because their outermost shell is full. In contrast, halogens, like chlorine (below to the right), have strong chemical reactivity because they lack an electron to fill the last electron shell.
Chemical reactivity also depends on the bonding forces between the atoms that make up the molecule. For new molecules to form, the bonds inside the molecules of the reagents must break. The greater the energy required, the less reactive the molecule.
Fluorine ( |F_{2}| ) is more reactive than oxygen ( |O_{2}| ) because the single bond between the two fluorine atoms is weaker than the double bond of oxygen gas.

Finally, the chemical reactivity of a substance depends on the energy balance of the reaction in which the substance is involved. This reaction energy corresponds to the difference between the energy required to break chemical bonds and the energy released during their formation.
Due to their electronic configuration, noble gases are chemically very stable. Indeed, as their outermost electron shell is saturated, noble gases do not seek to gain or lose electrons. Thus, they do not form chemical bonds with other atoms and are found as simple elements in nature.
Noble gases are generally used in applications where an inert medium must be created, that is to say, a medium which does not contain any substance capable of reacting chemically. This is the case in some applications such as fluorescent tubes, incandescent bulbs, and arc welding.
Five noble gas tubes in which an electric current flows.
A fuel is a flammable substance which can burn when exposed to an oxidizer.
The fuel can be solid, liquid, or gaseous. Combustible gases include hydrocarbons and hydrogen.
Butane, a type of hydrocarbon, is used as a fuel in furnaces that run on natural gas.
An oxidizer is a substance that causes combustion.
Most oxidizers are gaseous at ambient conditions. The most common oxidizer on Earth is oxygen. It is involved in most combustion reactions, whether they are fast, such as the explosion of gasoline vapours in an engine, or slow, such as in cellular respiration. Ozone is also an oxidizer, but it is very toxic. Fluorine and chlorine are oxidizers that belong to the halogen family. Their high reactivity makes them difficult to store.
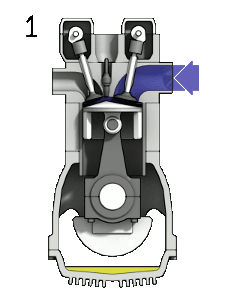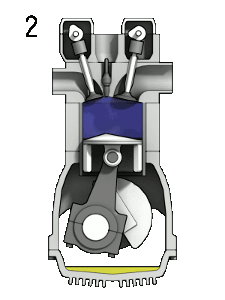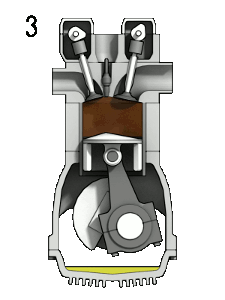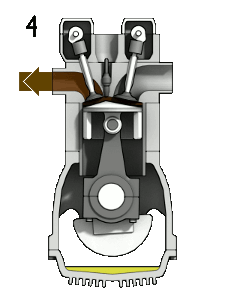
In a combustion engine, the oxygen in the air allows gasoline vapour to burn when the spark plug produces a spark.