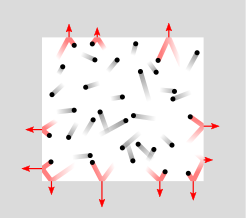
The pressure of a gas corresponds to the force it exerts on a surface due to the collisions of its particles on that surface.
All gases exert pressure on the surfaces they come into contact with. However, depending on whether the gas is atmospheric or contained in a container, the pressure exerted will not be measured in the same way.
Pressure corresponds to a force exerted per unit area. It can therefore be defined using the following mathematical formula:
|\displaystyle P=\frac{F}{A}|
where
|P| represents the pressure (in |{N/m^2}| or Pa)
|F| represents the force (in N)
|A| represents the area of the surface to which the force is applied (in |m^2|)
The pressure of a gas depends on the number of times its particles collide with an obstacle. The higher the number of collisions, the higher the pressure. However, under similar temperature conditions, particles in a light gas strike less hard and more often than particles in a heavy gas, which strike harder but less often. The sum of the forces exerted by the collisions is the same in both gases. This means that 1 mole of each gas under the same conditions exerts the same pressure, regardless of the size of its particles or its mass.
Representation of pressure as the result of collisions between the particles in a fluid contained in a container and the walls of the container.
Due to the Earth's gravitational force, gas particles in the atmosphere are more abundant at low altitudes than at high altitudes. These particles exert a force on all the objects they come into contact with. This force is called atmospheric pressure. It corresponds to the weight of the column of air above the surface that is subject to the force. It varies with altitude, temperature and weather conditions.
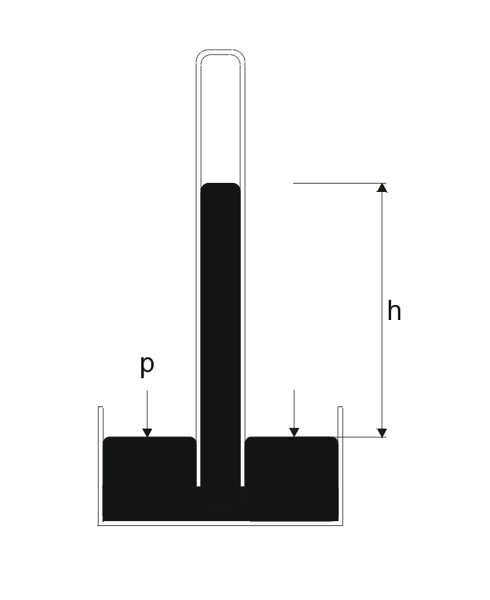
Atmospheric pressure is measured using a barometer. Originally designed by Evangelista Torricelli in 1643, this instrument consisted of a long glass tube filled with mercury (Hg) and inverted in a container containing the same metal. Gravitational force draws the mercury into the reservoir, while atmospheric pressure at the surface of the reservoir causes it to rise into the tube. The balance between the two forces determines atmospheric pressure.
Schematic Representation of the Torricelli Barometer
At sea level, the pitch of the mercury in the mercury tube is 760 mm Hg. This is normal atmospheric pressure. However, in the international system, pressure is measured in Pascal.
Equivalence of units of measurement for normal atmospheric pressure at sea level:
|\text {1 atm = 760 mm Hg = 101.3 kPa}|
The pressure gauge on a bottle of gas indicates a pressure of 350 kPa. What is the pressure of this gas in atmospheres? And in mm Hg?
In atmospheres:
|\displaystyle \frac{101.3\ kPa}{1\ atm}=\frac{350\ kPa}{x\ atm}|
|x=3.45\ atm|
The pressure is therefore equivalent to 3.45 atm.
In mm de Hg:
|\displaystyle \frac{101.3\ kPa}{760\ mm\ Hg}=\frac{350\ kPa}{x\ mm\ Hg}|
|x=2625.9\ mm\ Hg|
The pressure is therefore equivalent to 2625.9 mm Hg.
The barometer is only used to measure atmospheric pressure. When a gas is contained in a container, a manometer or pressure gauge is used instead.
A pressure gauge is a simple instrument that shows the pressure inside bicycle or car tyres (left). A manometer can also be used to determine the pressure of a compressed gas in a cylinder (right).
In U-tube manometers, the height of a column of liquid indicates the pressure of a gas. The liquid travels freely in the U-tube, which is connected to the container holding the gas. Despite the toxicity of mercury, this liquid is generally used in this type of manometer, as it reduces its size. It is the difference in level between the two columns of mercury in the U-tube that determines the pressure of the gas in the container. There are two types of U-tube manometer: open-end and closed-end.
Two Types of Manometer: Closed-end Manometer (left) and Open-end Manometer (right)
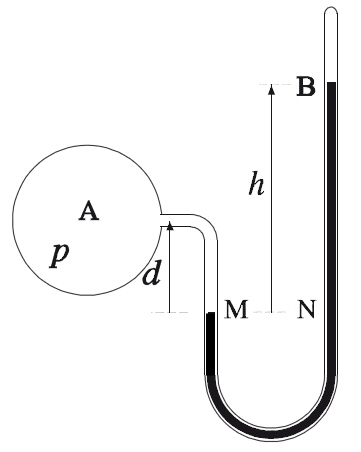
The pressure measured by a closed-end manometer actually corresponds to the gas pressure. So the difference in height in mm Hg between the two columns of liquid in the U-tube corresponds directly to the gas pressure.
What is the pressure of the gas in the balloon below?
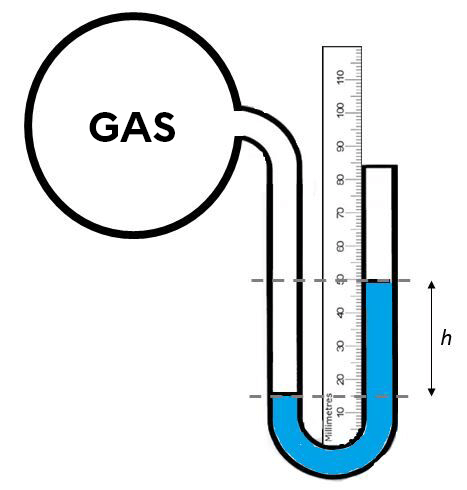
As the manometer has a closed end, the difference in height between the two columns of liquid in the U-tube gives the gas pressure directly.
Thus:
|50\ mm\ Hg -\ 15\ mm\ Hg =\ 35\ mm\ Hg|
The gas pressure is 35 mm Hg.
The pressure measured bye a open-end manometer is relative to the pressure exerted by the atmosphere in the open end of the manometer. The atmospheric pressure must therefore be taken into account when determining the gas pressure. In this way, the air and the gas exert a force on the surface of the liquid at each end of the tube, creating a kind of competition between the two.
- When the gas pressure is higher than the atmospheric pressure, the liquid level is lower on the gas side. The difference in height should then be noted and added to the atmospheric pressure.
- When the atmospheric pressure is higher than the gas pressure, the liquid level is higher on the gas side. The difference in height should then be noted and subtracted from the atmospheric pressure.
Actual pressure of a gas using an open-end manometer:
If |P_{gaz}| > |P_{atm}| then |P_{gaz}| = |P_{atm} + h|
If |P_{gaz}| < |P_{atm}| then |P_{gaz}| = |P_{atm} - h|
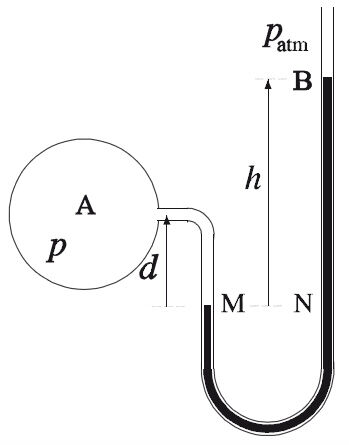
What is the pressure of the gas in the balloon below if the atmospheric pressure is 760 mm Hg?
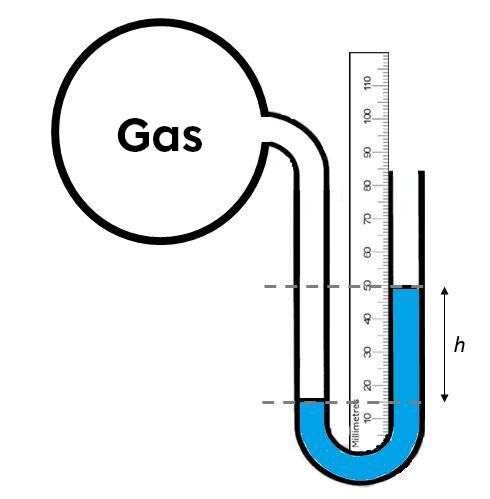
The manometer here is open-ended. It can therefore be seen that the pressure exerted by the gas is greater than the atmospheric pressure, since the liquid column is lower on the gas side. In this case, the difference in height of the liquid column must be added to the atmospheric pressure to obtain the gas pressure.
|P_{gaz} = P_{atm} + h|
|P_{gaz} = 760\ mm + (50\ mm - 15\ mm)|
|P_{gaz} = 795\ mm|
The pressure exerted by the gas is 795 mm Hg.