The ozone layer refers to the part of the atmosphere that contains a high concentration of ozone molecules |\text{O}_3| and partially absorbs the ultraviolet rays emitted by the Sun.
There are two types of ozone:
-
Stratospheric ozone forms a protective layer in the stratosphere and absorbs ultraviolet (UV) rays that can be harmful to human health.
-
Tropospheric ozone forms a layer in the troposphere and plays a role in the intensification of the greenhouse effect (GHG) by trapping greenhouse gases on the surface of the Earth. This ozone is also a component of smog.
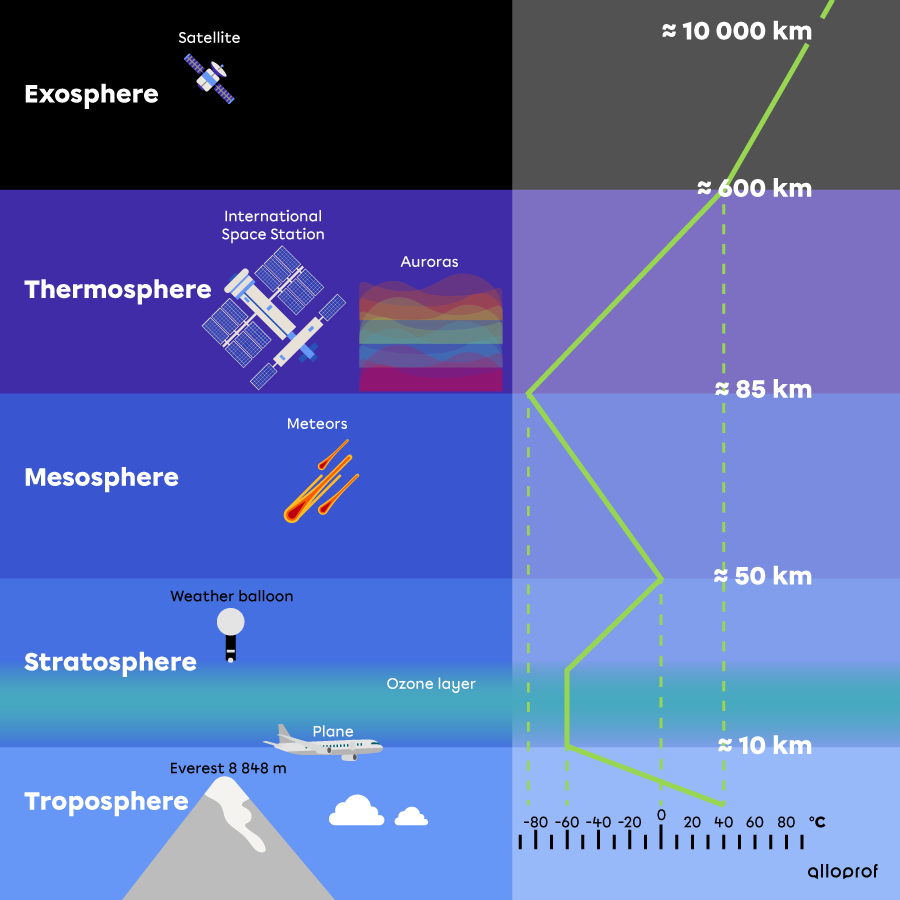
Without stratospheric ozone, life on the Earth’s surface would be impossible in the long term. Life could only exist deep in the oceans where UV rays cannot reach, since they can only penetrate the surface layer of the water.
The ozone in the troposphere enters our lungs and is harmful to our health. It is formed when nitrogen oxide molecules |\text{NO}_{x}| emitted in vehicle exhaust and factory fumes undergo chemical reactions under the effect of sunlight. Tropospheric ozone combines with other air pollutants, such as nitrogen dioxide |\text{NO}_{2}| and sulphur dioxide |\text{SO}_{2},| to form smog.
Smog is the haze of pollution that results from temperature inversion. The word smog is a portmanteau word made from the words smoke and fog.
When smog is present, the air in industrial cities can become unbreathable and have harmful effects on the population’s health. Certain at-risk people, including the elderly and people with emphysema or asthma, commonly experience increased breathing difficulties. Some people will experience eye irritation.
Tropospheric ozone is one of the components of smog and contributes to the intensification of the greenhouse effect. This phenomenon occurs when there is a significant increase in the concentration of GHGs in the atmosphere.
Atmospheric ozone is not distributed evenly around the Earth. It is mainly produced at the equator where the sunlight is more direct and intense. However, strong winds in the stratosphere carry this ozone away and towards the poles. The ozone layer is thicker in medium and high latitudes and thinner in the tropics.
However, in some places, the effect of chlorofluorocarbons (CFCs) has significantly reduced the thickness of the ozone layer.
Here are examples of products that contain CFCs:
| Aerosols | Spray paint, deodorants, insecticides, shaving cream, whipped cream |
| Foam insulation | Foams used in residential and commercial construction |
| Refrigerants | Air conditioning in commercial and residential building and cars, refrigerators, freezers |
| Cleaning products | Degreasers |
CFCs are responsible for the destruction of ozone molecules in the stratosphere. In 1987, 190 countries signed the Montreal Protocol, agreeing to reduce their CFC production. The ozone layer is now recovering, and ozone depletion events are becoming less frequent. Based on the most recent report issued in 2023, the layer is expected to recover by 2040 in some areas, by 2045 over the Arctic and by 2066 over the Antarctic.
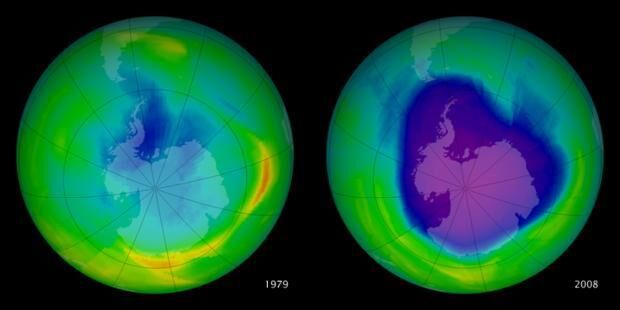
Difference in the ozone layer thickness over Antarctica between 1979 and 2008
When exposed to ultraviolet rays in the stratosphere, CFCs decompose and produce chlorine atoms. When these atoms interact with ozone, the ozone is decomposed.
Here is how it works:
-
UV radiation breaks down the CFC molecule, and a chlorine atom escapes.
-
The chlorine atom collides with an ozone molecule.
-
The chlorine atom captures one of the oxygen atoms found in ozone, producing chlorine monoxide and oxygen gas.
-
The chlorine monoxide then encounters a lone oxygen atom.
-
This interaction produces oxygen gas and frees the chlorine atom again, enabling it to interact with another ozone molecule. And the cycle repeats.
To rotate the molecules and view them from different angles, position the cursor over the animation, click and hold the left mouse button and move the cursor in the desired direction.
