An endothermic reaction is a transformation that absorbs energy.
An exothermic reaction is a transformation that releases energy.
Some chemical transformations absorb energy, while others release it. As thermal energy is generally the energy associated with chemical reactions, we speak of endothermic and exothermic reactions.
In an endothermic reaction, an input of energy is used to break the bonds that exist in the reactants, since these bonds are stronger than those in the products.
In an exothermic reaction, on the other hand, the bonds in the reactants are weaker than those in the products. As a result, more energy is released.
There are several ways of describing the energy involved in a reaction:
Examples of endothermic reactions
Melting snow (left); electrolysis of water (centre); cooking food (right)
Examples of exothermic reactions
Combustion a stick of incense (left); dissolution of NaOH (centre); redox reaction in a battery (right)
Both a physical transformation and a chemical reaction can involve an exchange of energy with the environment. Endothermic and exothermic reactions are therefore found in both physical and chemical reactions.
Physical transformations change neither the nature nor the characteristic properties of matter. The properties of matter are the same before and after the change.
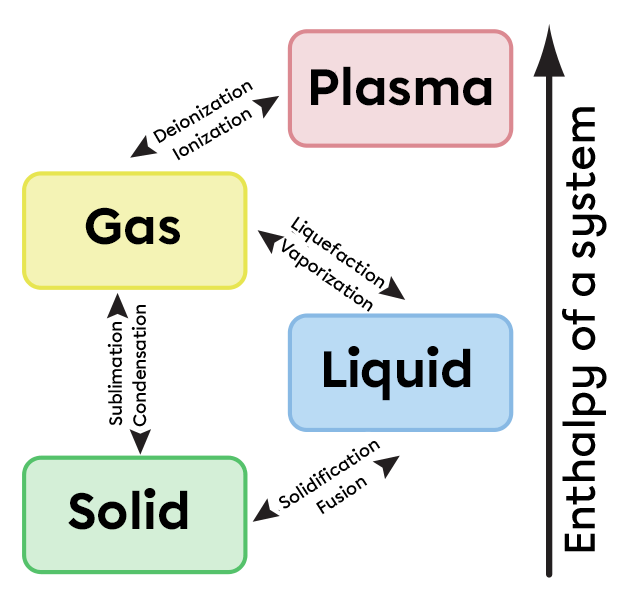
Changes in the phase of matter cause bonds between the particles that make it up to break or form. Some phase changes therefore absorb energy. Fusion, sublimation and vaporisation are endothermic reactions, since they require energy to reduce the forces of attraction between the particles. The particles will then separate further and tend more and more towards the gaseous state.
Conversely, exothermic phase changes are solidification, solid condensation and liquid condensation. In these cases, the attractions between the particles become greater and energy is released.
The enthalpy change involved in physical transformations can be represented graphically as in the following diagram:
Chemical transformations change the nature and characteristic properties of matter. New substances are formed as a result of the reaction.
During a chemical reaction, the molecules of the reactants break down and their atoms reorganise to form new molecules: the products. Generally speaking, breaking bonds requires energy, whereas forming bonds releases energy. However, it is the difference between the energy needed to break the reactants and that needed to form the products that determines whether a chemical reaction is endothermic or exothermic.
If more energy is needed to break chemical bonds than to form new ones, the reaction is endothermic. On the other hand, if the energy released is greater than that absorbed during the chemical reaction, the reaction is exothermic.
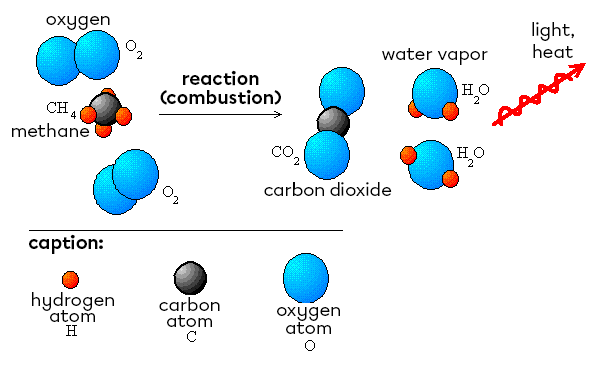
Example of an exothermic reaction: the combustion of methane
A reaction involving energy exchange can be expressed in the form of a thermochemical equation where the energy can be found on the reactants side (endothermic reaction) or on the products side (exothermic reaction).
Endothermic reaction
|\text{Reactants} + \text{energy} \rightarrow \text{Products} |
|H_{2}O_{(l)} + 44\ kJ \rightarrow H_{2}O_{(g)}|
Exothermic reaction
|\text{Reactants} → \text{Products} + \text{energy}|
|CH_{4(g)} + 2 O_{2(g)} → CO_{2(g)} + 2 H_{2}O_{(g)} + 802\ kJ|
The thermochemical equation can also be expressed in another way: by writing the enthalpy change next to the reaction. A positive enthalpy change indicates an endothermic reaction, while a negative enthalpy change indicates an exothermic reaction.
Endothermic reaction
|CaCO_{3(s)} → CaO_{(s)} + CO_{2(g)} \hspace {2 cm} \triangle H = 178\ kJ/mol |
Exothermic reaction
|4 Fe_{(s)} + 3 O_{2(g)}→ 2 Fe_{2}O_{3(s)} \hspace {2 cm} \triangle H = -824.2\ kJ/mol |
Using the thermochemical equation and the principles of stoichiometry, it is possible to carry out various calculations involving the quantities of matter and energy involved.
How much energy would be released by the complete combustion of 100.00 g of methane?
|CH_{4(g)} + 2 O_{2(g)} → CO_{2(g)} + 2 H_{2}O_{(l)} + 890\ kJ|
Solution
Using the periodic table, we find that 1 mole of |CH_{4}| has a mass of 16.05 g. Then, using a cross multiplication, we can find out how many moles 100.00 g corresponds to.
|\displaystyle \frac{16.05\ g}{1mol}=\frac{100.00\ g}{?}|
|? = 6.23\ mol|
As we know that the reaction releases 890 kJ for 1 mole of |CH_{4}|, a second cross multiplication allows us to find how much energy is released for 6.23 mol.
|\displaystyle \frac{890\ kJ}{1\ mol}=\frac{?}{6.23mol}|
|? = 5544.7\ kJ|
So the complete combustion of 100.00 g of methane gives off 5544.7 kJ.