An endothermic reaction is a reaction that absorbs energy from the environment, thereby lowering the energy content of the medium.
When a chemical reaction absorbs heat, the temperature of the surrounding environment decreases. The final temperature is then lower than the initial temperature. It is therefore the medium that is responsible for this transfer of energy.
An endothermic reaction can be recognised in various ways:
An endothermic reaction is recognised when, in a chemical equation, the associated energy value (or thermal effect) is integrated on the reactant side of the equation.
The equation for an endothermic reaction is of the type:
|\text{Reactants} + \text{Energy} \rightarrow \text{Products}|
The fusion of water is an endothermic reaction:
|H_{2}O_{(s)} + 6 kJ \rightarrow H_{2}O_{(l)}|
The decomposition of ammonia is an endothermic reaction:
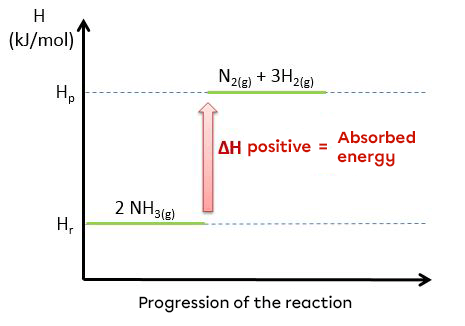
|2 NH_{3(g)} + 95.4 kJ \rightarrow N_{2(g)} + 3 H_{2(g)}|
The enthalpy (H) of a molecule is measured in joules per mole (J/mol ) or kilojoules per mole (kJ/mol). In an endothermic reaction, the total energy of the reactants, or the total enthalpy (H), is less than that of the products. The change in enthalpy (ΔH) is therefore positive.
For an endothermic reaction:
|H_{r} < H_{p}|
|\text{Reactants} \rightarrow \text{Products} \hspace {2 cm} \triangle H = \text {positive value}|
Here are two examples of endothermic reactions:
|CaCO_{3(s)} \rightarrow CaO_{(s)} + CO_{2(g)} \hspace {2 cm} \triangle H = \text {+ 178 kJ/mol}|
|2 NH_{3(g)} \rightarrow N_{2(g)} + 3 H_{2(g)} \hspace {2 cm} \triangle H = \text {+95.4 kJ/mol}|
Here is a diagram showing the endothermic reaction for the decomposition of ammonia and the energy evolution during this reaction.
When bonds are broken in a molecule, energy is always absorbed in the form of heat. This stage is always endothermic.
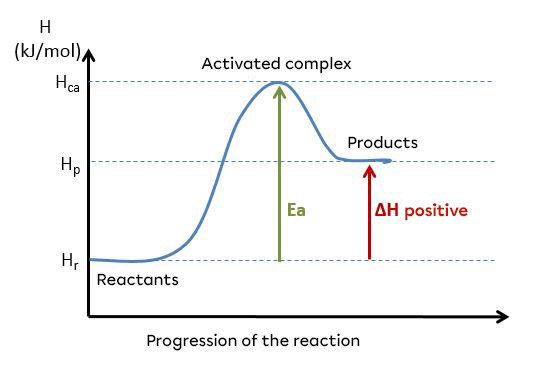
A reaction can be said to be endothermic when the direct activation energy (|E_{ad}|) is greater than the reverse activation energy (|E_{arev}|). These energies can be illustrated using an energy diagram.
For an endothermic reaction:
|E_{ad} > E_{arev}|
There are several examples of endothermic reactions, including the majority of chemical decompositions, whether caused by heat, light or electricity (electrolysis).
Example of an energy diagram for an endothermic reaction