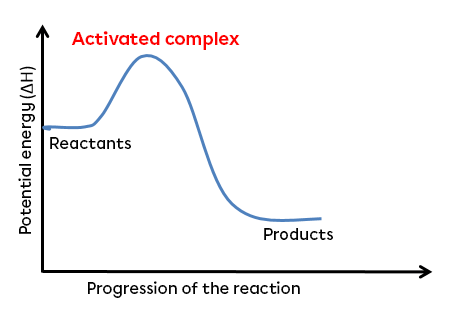
The activated complex is an unstable, energetic transition state formed during the transformation of reactants into products.
The activation energy is the minimum amount of energy required to initiate a chemical reaction, whether endothermic or exothermic.
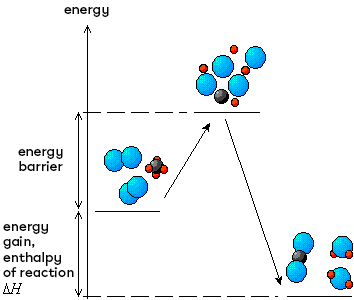
For a chemical reaction to take place, the reactant molecules must collide. According to collision theory, certain conditions are necessary for a reaction to take place. The collision of the reactant molecules must be efficient and not elastic, so that the activated complex can be formed. It must therefore take place with a minimum of energy, known as the activation energy. These two pieces of information can be found in the energy diagram of a reaction.
Before the reactant molecules are transformed into products, a highly energetic complex intermediate molecule called the activated complex is formed. So, for a very brief moment, the reactants' bonds weaken while new bonds begin to form. This transitional state is only temporary, as it is highly unstable. In fact, this intermediate grouping of atoms is virtually undetectable as the atoms tend to rapidly reform molecules in order to regain stability. Because of its instability, the activated complex always has the maximum enthalpy of a reaction. It is therefore always at the top of the curve in an energy diagram.
The activated complex always lies between the reactants and the products and at the top of the curve in an energy diagram.
The formation of the activated complex results from the efficient collision of reactant molecules. Although some of the reactant bonds are still present while some of the products are being formed, this transition state is often considered to be the brief instant during which all the chemical bonds have disappeared. It is therefore symbolised by a cluster of unbound atoms that will reorganise to form new molecules.
The activated complex is often represented as a group of chemically unbound atoms. In the graph below illustrating the combustion of methane, we can see that the chemical bonds of the reactants are completely broken before new ones are formed in the products.
For the reactants to reach the activated complex state, their collisions must take place with a minimum of energy, which corresponds to the activation energy.
The formation of the activated complex involves the rearrangement of the attractive forces between the atoms of the reactants so that they can be transformed into products. This rearrangement requires a very large amount of energy. So, for a chemical reaction to occur, the reactants must receive sufficient energy to form the activated complex. This minimum level of energy required to reach the transition state is called the activation energy and is symbolised by Ea. Its value corresponds to the energy difference between the activated complex and the reactants.
The mathematical formula for calculating the activation energy of a direct reaction is as follows:
Ea=Hac−Hr
Take the graph below:
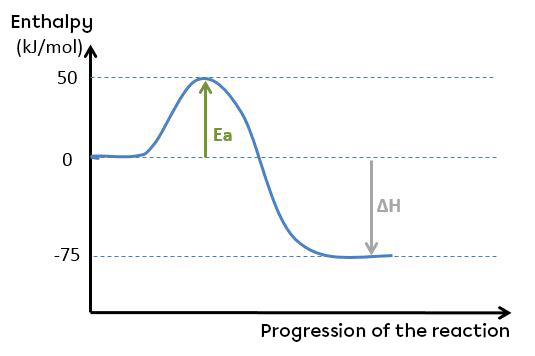
The activation energy can be calculated as follows:
Ea=Hac−Hr
Ea=50−0
Ea=50 kJ/mol
We can also talk about the activation energy of the reverse reaction (Ea rev ). This is the value corresponding to the variation between the enthalpy of the products and that of the activated complex. This energy corresponds to the activation energy in the reverse reaction (the products are then the reactants and the reactants become the products).
To calculate the activation energy of a reverse reaction, use the following mathematical formula:
Ea rev=Hac−Hp
Take the graph below:
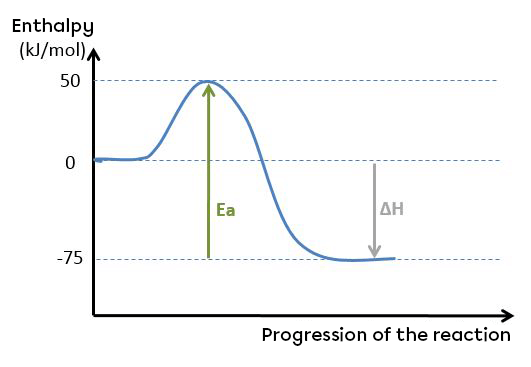
The activation energy of the reverse reaction can be calculated as follows:
Ea rev=Hac−Hp
Ea rev=50−(−75)
Ea rev=125 kJ/mol
Activation energy can also be represented using the graph showing the number of molecules as a function of kinetic energy. In this case, the energy barrier corresponds to the activation energy. All the molecules with this minimum energy or more will collide effectively and will be able to react.
The molecules that make up the reactants must therefore have sufficient energy to be able to break the forces or bonds that hold them together, in order to form new bonds and form new molecules. This minimum energy corresponds to the activation energy. The purple area in the graph below corresponds to all the reactant molecules that have sufficient energy to react.
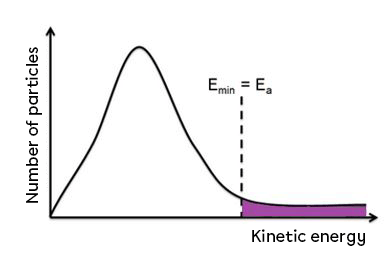
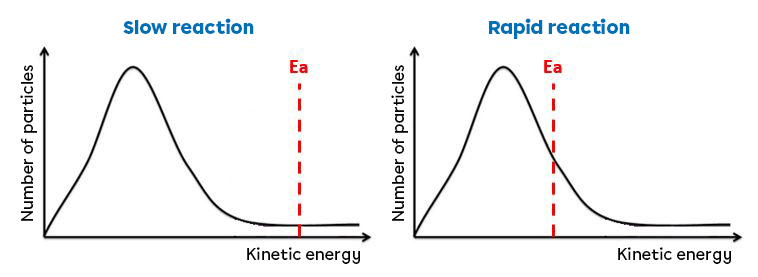
The value of the activation energy can help us to estimate the reaction rate of a reaction. A high activation energy value indicates a slow reaction. In the case of a slow reaction, few reactants (the area under the curve to the right of the activation energy) have sufficient kinetic energy to cross the barrier and form new products. Conversely, a low activation energy value indicates a fast reaction.
The graph on the left illustrates a relatively slow reaction, since the activation energy is high, whereas the graph on the right illustrates a rapid reaction, since the activation energy is low.
Certain factors can influence the minimum energy level required to initiate a reaction: