In order to fully understand the acidity and basicity constants, it is important to adjust our definition of the terms "acid" and "base" a little. Usually, they are described by their macroscopic properties and molecular structure. They are then defined according to the ions they produce. However, this definition has certain limitations since it does not provide a satisfactory explanation of their behaviour. In an effort to overcome these limitations and better define these substances, various theories have been developed:
According to Arrhenius theory:
- An acid is a substance which, when brought into aqueous solution, releases hydrogen ions |(H^{+})|.
- A base is a substance which, when brought into aqueous solution, releases hydroxide ions |(OH^{-})|.
In 1887, the Swedish chemist Svante Arrhenius developed the first theory on acids and bases. This theory forms the basis of our current definition of acids and bases. Developed from the observation of the ions that these substances tend to form, an acid is defined as a substance that produces |(H^{+})| ions in solution, while a base produces |(OH^{-})| ions.
| Acids that dissociate in water and the resulting ions | Bases that dissociate in water and the resulting ions |
| |HBr_{(aq)} \rightarrow H^{+}_{(aq)} + Br^{-}_{(aq)}| | |LiOH_{(aq)} \rightarrow Li^{+}_{(aq)} + OH^{-}_{(aq)}| |
| |H_{2}SO_{4(aq)} \rightarrow H^{+}_{(aq)} + HSO^{-}_{4(aq)}| | |KOH_{(aq)} \rightarrow K^{+}_{(aq)} + OH^{-}_{(aq)}| |
| |HClO_{4(aq)} \rightarrow H^{+}_{(aq)} + ClO^{-}_{4(aq)}| | |Ba(OH)_{2(aq)} \rightarrow Ba^{2+}_{(aq)} + 2\; OH^{-}_{(aq)}| |
Arrhenius theory provides a simple description of acids and bases and is useful in describing their electrolytic dissociation. However, in order to comply with this theory, the molecular structure of an acid must contain at least one hydrogen atom as a source of hydrogen ions, while that of a base must contain at least one oxygen atom and one hydrogen atom to generate a hydroxide ion. However, we sometimes notice certain basic behaviours (such as turning red litmus paper blue) in molecules that do not comply with this structure. So, although simple and practical, Arrhenius theory has certain limitations:
- It does not make it possible to explain the formation of |(H_{3}O^{+})| hydronium ions formed by the attraction of water and a hydrogen ion during the electrolytic dissociation of an acid. In fact, the |H^{+}| aqueous ion hardly exists in aqueous medium since it tends to react with the water molecule due to its polarity.
- It does not make it possible to explain the basic behaviour of certain substances that do not contain atoms necessary for the production of hydroxide ions, as in the case of |NH_{3}| or |CaCO_{3}|.
- It does not make it possible to explain reactions between acids and bases which occur in non-aqueous mediums.
According to the Brønsted-Lowry theory:
An acid is a substance capable of donating one or more protons in the form of a |H^{+}| hydrogen ion to another substance: it is a proton donor.
A base is a substance capable of receiving one or more protons in the form of a |H^{+}| hydrogen ion coming from another substance: it is a proton acceptor.
A hydrogen ion is a hydrogen atom that has lost its single electron. It only has one proton left in the nucleus. This is why, in the box above, we refer to proton donor and proton acceptor.
In 1923, chemists Johannes BrØnsted and Thomas Lowry developed a theory on acids and bases to solve the problems posed by Arrhenius theory. In this theory, the emphasis is placed on the roles that acids and bases have in a chemical reaction rather than on their properties in aqueous solutions.
As in Arrhenius theory, the molecular formula of an acid according to BrØnsted-Lowry must contain a hydrogen atom. However, any negative ion can play the role of base to accept the proton yielded by the acid. In other words, since this theory is based on the transfer of a proton rather than on the structure of the molecules involved, each acid releasing a proton must have a base capable of receiving that proton.
The term “conjugate acid of a base” is used to refer to the particle formed by the base which has captured a proton, while the particle remaining when the acid has lost a proton is called “conjugate base of an acid.”
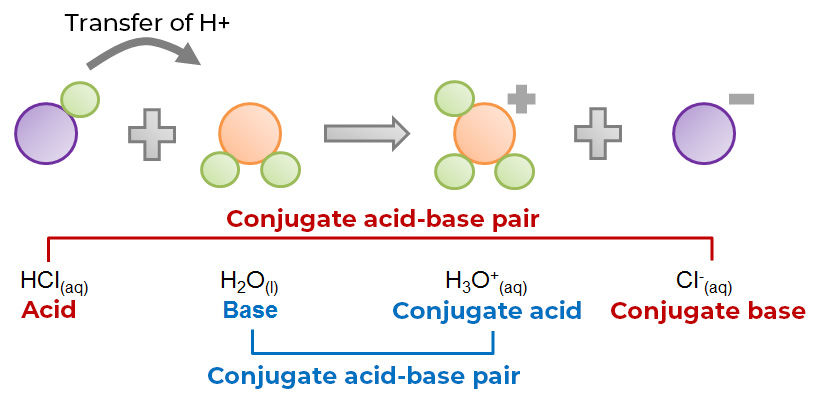
Reaction between hydrochloric acid and water according to BrØnsted-Lowry theory:
Reaction between ammonia and water according to BrØnsted-Lowry theory:
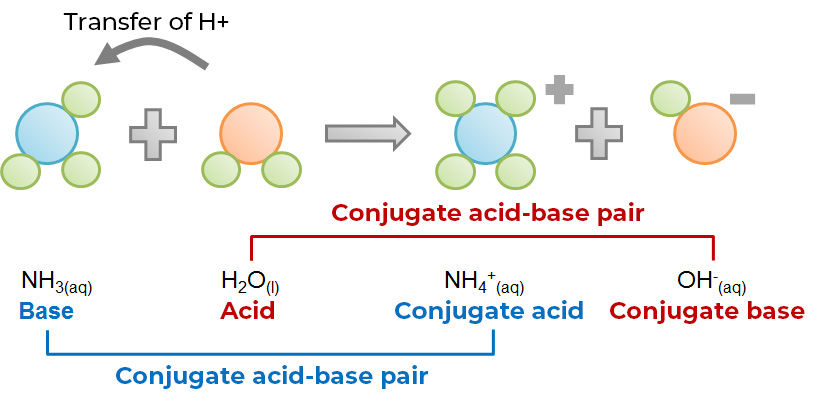
According to this theory, any substance can act as a base as long as another substance behaves like an acid at the same time. Also, a substance cannot be classified as an acid or a base depending only on a given reaction. In fact, depending on the reaction considered, the same substance can sometimes play the role of an acid, and at other times a base. This is also the case with the water molecule in the two examples above. When hydrochloric acid is present, water is a base since it will receive the proton lost by the acid. However, in the presence of ammonia, water acts as an acid due to the loss of a proton to ammonia. Similar to water, such a substance, which can play both roles depending on the reaction considered, is known as an amphoteric substance.