The laboratory report, commonly referred to as the lab report, is a written document that presents a summary of the experiment and the results that are obtained.
Although the content of a lab report follows a general structure, it may vary depending on your grade level, your teacher's requirements and the type of lab you are doing. Make sure you follow the instructions when completing your lab report.
Conclusion
When writing a lab report, the text should be presented in a particular way. There are different formatting standards depending on the teacher's instructions. Here are some key points to get you started.
The colour, size and style of font
If the lab report is handwritten, consider which type of writing utensil to use. For example, can it be written in lead pencil or blue ink?
If the lab report is written using an electronic device (e.g., computer, tablet), think about the choice of font, its size and colour. Often, the report is requested to be written in 12 point Times New Roman black font.
Text alignment and spacing
Generally, the text of a lab report written in a word processor should be justified, meaning that the width of the paragraphs should be set. In addition, it is important to check whether the report should be single or double spaced. Usually, if the text is entered in a word processor, the instruction is to select 1.5 or 2 line spacing.
Page numbers
If the pages of the laboratory report are to be numbered, the pagination rules specified by the teacher must be followed.
A lab report should be objective, meaning that it should not contain any personal judgements. It should not be possible to guess the writer's personal opinions when reading it.
To this end, a lab report is usually written in a passive voice. It is recommended to avoid the pronouns I and we.
| Preferred phrasing | Phrasing to avoid |
|---|---|
|
The obtained results are [...]. This example is phrased in the passive voice. It does not refer to the person(s) conducting the experiment or writing the report. This is the preferred style. |
I obtained the results [...]. The phrasing used in these examples should be avoided because the pronouns I and we replace the person(s) writing the report. |
In addition, descriptive language that is open to interpretation should be avoided in lab reports. Instead, neutral and precise language should be used.
| Preferred phrasing | Phrasing to avoid |
|---|---|
|
The mass of Substance 1 is 2 kg. These examples are phrased using objective and precise language. They do not express an opinion or a subjective description. |
The mass of Substance 1 is huge. This example is phrased using a subjective description. The interpretation of this statement may differ from one person to another. |
The use of the word find should be avoided. It can always be replaced by more precise terms such as measure, calculate, determine, etc.
For example, instead of writing "Objective: To find the boiling point of water," it is better to write "Objective: To determine the boiling point of water." Similarly, instead of writing "Find the temperature of water," it is better to write "Measure the temperature of water."
The title page is the cover page of the lab report. It usually includes the following information:
-
Your name and the names of your teammates, if applicable, in alphabetical order
-
Your group number
-
The title of the lab report
-
The name of your teacher after the phrase “Report submitted to … ” or “Work presented to … ”
-
Your grade and subject
-
The name of your school
-
The date of submission of the report
The layout of the title page may vary depending on the standards at your school. Some information may be required to be capitalized, italicized, bolded, centered, right aligned, etc. It is important to ensure that you respect the specific requirements.

The introduction is the first section of the lab report. It includes the objective(s), the background research and the hypothesis. Sometimes it also contains the context of the experiment, which is usually provided by the teacher and included at the very beginning of the report, before the objective(s).
The objective of the experiment, sometimes called the purpose, is a statement that explains what is to be achieved in the lab.
It should begin with a verb in the infinitive and is usually written in one sentence, unless there are several objectives.
Objective: To determine the effect of decreasing the temperature from 20°C to -10°C on the state of the water.
Objective: To determine the relationship between the potential difference across a resistor and the current flowing through it.
The background research is the section where the theoretical concepts relevant to the lab are laid out. In other words, it is the information that must be known in order to be able to carry out the experiment and meet the objective. It is used as a reference throughout the lab. It should be written in complete sentences.
Depending on the level and requirements, it may include definitions, formulas, reference values, independent and dependent variables, units of measurement and other information.
In a lab where the objective is to determine the effect of decreasing temperature from 20 °C to -10 °C on the state of water, the following information would be included in the background research:
The freezing point of water is at 0 °C. When the temperature of water is below 0 °C, it is in solid form. When the temperature of water is above 0 °C, it is in liquid form.
In a lab where the objective is to identify the acidic or basic nature of a solution, the following information would be included in the background research:
Bromothymol blue is an acid-base indicator. It turns yellow when the pH is between 1 and 6, green when the pH is between 6 and 7.5 and blue when the pH is between 7.5 and 14.
A substance with a pH lower than 7 is acidic. A substance with a pH greater than 7 is basic.
The hypothesis is a proposed outcome to the objective. Since the experiment has not yet been carried out, it is normal not to know the outcome with certainty. It is a supposition made based on observations, on what is already known or on the background research. The hypothesis has to be justified.
In a lab where the objective is to determine the most insulating material between wool, plastic and aluminum foil, the hypothesis could be the following:
Wool would be the most insulating material because it is the most porous material.
In a lab where the objective is to determine whether mass is conserved in a chemical reaction, the hypothesis might be as follows:
The mass of the reactants should be equal to the mass of the products because in a chemical reaction the atoms rearrange to form new substances, but the number of atoms is conserved.
The body is the main section of the lab report. It includes the materials and equipment, procedure, results and analysis.
The materials and equipment to be used in the experiment are listed in point form in one or more columns. The instruments, tools and substances required for the experiment should be included. The quantity and capacity of the instruments should be specified, if applicable. The concentration of the substances used should be specified, if applicable. Safety equipment is sometimes included.
If necessary, you can refer to the lab equipment concept sheet for pictures and names of instruments, glassware and safety equipment.
It is not necessary to name materials used in writing the lab report (e.g., pencil or eraser) or materials for storage and cleaning, because these items are not used during the actual lab. A person who wants to replicate the experiment would not need these materials to repeat the procedure.
This is the materials list for an experiment where the boiling point of water is measured:
-
1 hot plate
-
1 universal stand
-
1 thermometer clip
-
1 thermometer
-
1 x 500 mL beaker
-
1 pair of beaker tongs
-
1 wash bottle
-
Distilled water
This is the materials list for the acid-base neutralization of an HCl solution with a NaOH solution:
-
1 x 100 mL graduated cylinder
-
1 x 50 mL burette
-
1 burette stand
-
1 universal stand
-
1 x 125 mL Erlenmeyer flask
-
1 x 500 mL beaker
-
HCl solution of unknown concentration
-
NaOH solution of 1 mol/L concentration
-
Bromothymol blue
The procedure consists of the steps to be taken in order to carry out the experiment. The procedure is sometimes called the protocol or the methods. The procedure must be detailed and precise so that anyone can repeat the experiment exactly as it was done before. It is particularly important to specify the amounts of substances and the capacities of the glassware.
The procedure steps should be numbered and presented in the order in which they will be carried out. In addition, they must begin with an imperative verb, like direct instructions. If several steps are to be repeated, their numbers should be specified: "Repeat steps X to Y."
Basic operations such as "Put on safety goggles" or "Put away the materials" do not need to be included. Only the specific steps of the experiment should be listed.
In a lab where the mass of different granular solids is to be measured, the procedure would be as follows:
-
Measure the mass of the empty weighing dish using the scale and record it.
-
Place Solid A to be weighed in the dish.
-
Place the dish containing Solid A on the pan of the scale.
-
Measure the mass of the dish with Solid A and record it.
-
Repeat steps 2 to 4 with solids B, C and D.
Set-up diagram
If a specific set-up is required in the procedure, a diagram of the set-up should be included. The diagram should be clear and the components should be identified by name or symbol so that anyone can repeat the same experiment.
The diagram of the set-up is usually inserted after the procedure step that mentions it or at the very end of the procedure. The diagram should be numbered and have a title.
To find out how to make a scientific diagram, consult the following concept sheet.
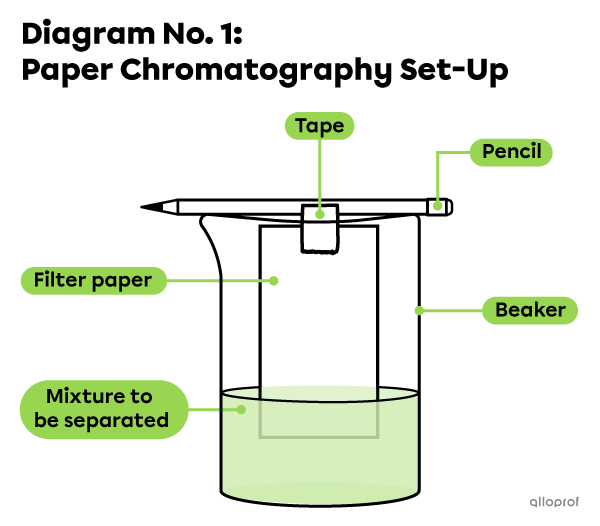
In a lab where spinach leaf pigments are separated from a mixture of spinach and isopropyl alcohol, the set-up diagram might look like this:
Procedure
- Construct the following set-up (Diagram No. 1) by taping the filter paper strip to the pencil.
The results are the measurements and observations collected during the experiment. They are usually presented in text form accompanied by drawings, diagrams, tables, graphs and/or sample calculations. The content of this section varies according to the experiment carried out and the type of results obtained.
The presentation of the results is done in a logical order. For example, if a graph is made based on the measured data, the table of data is presented first, followed by the graph. If results are calculated from the measured data, the data table is presented first, followed by the sample calculations and the results table.
Scientific drawings and diagrams
In some cases, observations cannot be written down as numerical values or simple descriptions. Sometimes it is necessary to make a drawing or a diagram. For example, in a lab where observations are made under a microscope, scientific drawings have to be made. Diagrams and drawings should be titled and numbered according to the order in which they appear in the lab report.
To find out how to make a scientific drawing or diagram, consult the following concept sheet.
Tables
Tables are used to present observations, measurements or results in an organized manner. There are two types of tables: the data table and the results table. The data tables group data that have been observed or measured directly in the lab. The results tables, on the other hand, contain results that are calculated or deduced from the data collected in the lab.
Data tables and results tables should be numbered according to the order in which they appear in the lab report and should have a title representative of their content.
To find out how to create tables, consult the following concept sheet.
Secondary 4-5
Depending on the teacher's requirements, it may be necessary to indicate the uncertainties of the measured values in the results tables and to respect the number of significant figures.
Graphs
Graphs are used to illustrate a relationship between two variables. Graphs should be numbered according to the order in which they appear in the lab report and should have a title that is representative of their content. They should be large enough to see their content, but small enough to fit on the page. Graphs can be made by hand or with software, depending on the requirements.
To find out how to make a graph, see the following concept sheet.
Secondary 4-5
Depending on the teacher's requirements, it may be necessary to respect the significant figures or to represent the uncertainty of each variable around the points on the graph.
Examples of calculations
If results have been obtained using calculations, these calculations should be demonstrated.
If the calculations are done only once, they should be presented in a section entitled Calculations. This shows all the calculations that led to the results. If identical calculations are performed several times, but with different values, they should be presented in a section entitled Examples of calculations. Only one example of each type of calculation should be shown.
To present the calculations, identify what is being calculated, the values and units, the formula(s) to be used, the solution and the result.
Secondary 4-5
Depending on the teacher's requirements, significant figures or uncertainty calculations may be required.
In a lab where the volume of a solid is to be measured by displacement of water, the calculation can be written as follows:
Calculation
The volume of the unknown solid |(V_\text{solid})|
|\begin{align}V_\text{water} &= 50\ \text{mL}\\V_\text{water + solid}&= 68\ \text{mL}\\\\
V_\text{solid}&=V_\text{water + solid}-V_\text{water}\\V_\text{solid} &= 68\ \text{mL}-50\ \text{mL}\\V_\text{solid} &= 18\ \text{mL}\end{align}|
In a lab where the density of different substances has to be calculated from their mass and volume measurements, the calculation for Substance 1 can be written as follows:
Example of a calculation
The density |(\rho)| of Substance 1
|\begin{align} m_1 &= 10\ \text{g}\\
V_1 &= 12\ \text{mL}\\\\
\rho_1 &= \dfrac{m_1}{V_1}\\\\
\rho_1 &= \dfrac{10\ \text{g}}{12\ \text{mL}}\\\\
\rho_1 &\approx 0.83\ \text{g/mL}
\end{align}|
The analysis is the most important section of the lab report. It explains the important results relevant to the objective(s) of the lab and provides feedback on the experimental approach. It is presented as a narrative text.
Analysis of results
The analysis of results is a section where the important results of the experiment are presented and explained with links to the theory included in the background research. Values obtained can be compared with each other or with theoretical values. Similarities, differences or trends are highlighted in order to meet the objective(s) of the lab.
In a lab where the most insulating material needs to be determined, the analysis could be as follows:
The test tubes were immersed in a beaker of ice for 2 minutes. The water in the wool-covered test tube dropped from 20°C to 17°C, which corresponds to a decrease of 3°C. The water in the paper-covered test tube decreased from 20°C to 12°C, which corresponds to a decrease of 8°C. The water in the uncovered test tube decreased from 20°C to 9°C, which corresponds to a decrease of 11°C. The most insulating material is therefore wool, since it resulted in the smallest temperature drop. This result is explained by the fact that wool is a porous material that contains a large amount of trapped air. Air is a thermal insulator.
Sources of error and process analysis
The sources of error are the reasons why the results are not perfectly accurate. They can be related to the experimental approach, the execution of the procedure, the materials and equipment, an environmental factor in the laboratory space (light, temperature, pressure, etc.) or human error (reaction time), for example.
The identified sources of error should be consistent with the result obtained. For example, if the result obtained is smaller than the theoretical value, the sources of error must explain that smaller result.
Identifying the sources of error then allows you to analyze the experimental approach and propose improvements.
Possible sources of error are:
-
The sample substance is too small to allow an accurate measurement of the mass.
-
The difference between the values of two consecutive gradations of the graduated cylinder was too high to allow an accurate measurement of the volume of the sample.
-
The reaction time when the stopwatch is used results in time values that are too large.
Possible improvements to the experimental approach include:
-
Using a larger sample of the substance would allow a more accurate measurement of mass.
-
Using a graduated cylinder with a smaller difference between the values of two consecutive gradations would allow a more accurate measurement of the volume of the sample.
-
Using an automatic timing device to measure the elapsed time would result in a more accurate measurement.
The conclusion is the last part of the lab report. It consists of a simple summary of the lab. It is used to say whether the objective was achieved by reiterating the important results. It usually contains the following parts:
-
Return to the objective(s)
Each objective of the lab is restated and answered using the important results. It is confirmed whether each objective was achieved.
-
Return to the hypothesis
The hypothesis is restated and it is explained whether the hypothesis is supported or rejected based on the relevant results.
-
Discussion
The discussion is the final part of the conclusion. Depending on the nature of the lab and what is required, the discussion can take different forms. For example, this section could include improvements to the experimental approach in order to reduce the sources of error, future experiment suggestions to push the research further or concrete applications of the results obtained (Why is it useful to know these results?).
In a lab where the boiling point of water has to be determined, the conclusion could be as follows:
The objective of the lab was to determine the boiling point of water. The result obtained is 99°C. The objective was achieved. The hypothesis was that it would be 100°C, which is the known theoretical value. The result is close to the theoretical value, so the hypothesis is supported. Since the boiling point is a characteristic property, knowing the boiling point of water makes it possible to identify whether an unknown substance is water by comparing its boiling point with that of water.
If the lab report contains data from a publication (book, website, article, etc.), this should be listed in the Reference section. This section is found at the end of the report.
There are several ways to write references. It is also sometimes necessary to use footnotes. Make sure you follow the specific requirements.
If a document is referred to that is too extensive to be included in the lab report, it should be included as an appendix. This can be a protocol provided by the teacher, a set of theoretical data, graphs, diagrams, etc.
Are you participating in the Science Fair and have to write a lab report? On this page you can find tools to help you with this task, including the Report Writing Guide.